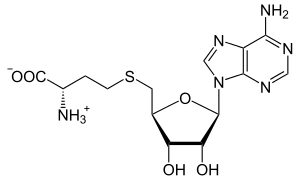
S-Adenosyl-L-homocysteine (SAH) is the biosynthetic precursor to homocysteine.[1] SAH is formed by the demethylation of S-adenosyl-L-methionine.[2][3] Adenosylhomocysteinase converts SAH into homocysteine and adenosine.
 | |
| Names | |
|---|---|
| IUPAC name S-(5′-Deoxyadenos-5′-yl)-L-homocysteine | |
| Systematic IUPAC name (2S)-2-Amino-4-({[(2S,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methyl}sulfanyl)butanoic acid | |
| Other names AdoHcy, 2-S-adenosyl-L-homocysteine, 5′-S-(3-Amino-3-carboxypropyl)-5′-thioadenosineS-adenosylhomocysteine, SAH | |
| Identifiers | |
3D model (JSmol) | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.012.328 |
| KEGG | |
| MeSH | S-Adenosylhomocysteine |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C14H20N6O5S | |
| Molar mass | 384.41 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Biological role edit
DNA methyltransferases are inhibited by SAH.[4] Two S-adenosyl-L-homocysteine cofactor products can bind the active site of DNA methyltransferase 3B and prevent the DNA duplex from binding to the active site, which inhibits DNA methylation.[5]
References edit
- ^ Finkelstein JD (2000). "Pathways and regulation of homocysteine metabolism in mammals". Seminars in Thrombosis and Hemostasis. 26 (3): 219–225. doi:10.1055/s-2000-8466. PMID 11011839.
- ^ Ribbe MW, Hu Y, Hodgson KO, Hedman B (April 2014). "Biosynthesis of nitrogenase metalloclusters". Chemical Reviews. 114 (8): 4063–4080. doi:10.1021/cr400463x. PMC 3999185. PMID 24328215.
- ^ James SJ, Melnyk S, Pogribna M, Pogribny IP, Caudill MA (August 2002). "Elevation in S-adenosylhomocysteine and DNA hypomethylation: potential epigenetic mechanism for homocysteine-related pathology". The Journal of Nutrition. 132 (8 Suppl): 2361S–2366S. doi:10.1093/jn/132.8.2361S. PMID 12163693.
- ^ Kumar R, Srivastava R, Singh RK, Surolia A, Rao DN (March 2008). "Activation and inhibition of DNA methyltransferases by S-adenosyl-L-homocysteine analogues". Bioorganic & Medicinal Chemistry. 16 (5): 2276–2285. doi:10.1016/j.bmc.2007.11.075. PMID 18083524.
- ^ Lin CC, Chen YP, Yang WZ, Shen JC, Yuan HS (April 2020). "Structural insights into CpG-specific DNA methylation by human DNA methyltransferase 3B". Nucleic Acids Research. 48 (7): 3949–3961. doi:10.1093/nar/gkaa111. PMC 7144912. PMID 32083663.
External links edit
🔥 Top keywords: Akademia e Shkencave e RPS te ShqiperiseAlexandria Ocasio-CortezBilderberg GroupCristiano RonaldoDong XiaowanMinecraftOperation GladioPrimal cutRiot FestStrictly Come Dancing (series 7)Main PageSpecial:SearchUFC 302Wikipedia:Featured picturesChloe BennetIslam MakhachevDeontay WilderZhilei ZhangCarlo AncelottiDustin PoirierFranz Kafka2024 Indian general election2024 ICC Men's T20 World CupCleopatraGodzilla Minus OneRob BurrowList of European Cup and UEFA Champions League finalsDeaths in 2024UEFA Champions LeagueToni KroosMichael CrichtonJude BellinghamYouTubeAngelina JolieClaudia SheinbaumFuriosa: A Mad Max SagaReal Madrid CF2024 South African general election2019 Indian general electionDonald TrumpSean StricklandCivil War (film)Sikkim Krantikari MorchaList of most-subscribed YouTube channelsAaron Jones (cricketer)Eric (miniseries)The First OmenPapua New GuineaEminem
