Betulin is an abundant, naturally occurring triterpene. It is commonly isolated from the bark of birch trees. It forms up to 30% of the dry weight of silver birch bark.[2] It is also found in birch sap.[citation needed] Inonotus obliquus contains betulin.[3]
 | |
| Names | |
|---|---|
| IUPAC name Lup-20(29)-ene-3β,28-diol | |
| Systematic IUPAC name (1R,3aS,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bR)-3a-(Hydroxymethyl)-5a,5b,8,8,11a-pentamethyl-1-(prop-1-en-2-yl)icosahydro-1H-cyclopenta[a]chrysen-9-ol | |
| Other names Betulinol, betuline, betulol, betulinic alcohol, trochol | |
| Identifiers | |
3D model (JSmol) | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.006.797 |
| EC Number |
|
| KEGG | |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C30H50O2 | |
| Molar mass | 442.728 g·mol−1 |
| Appearance | solid with needle-like crystals[1] |
| Melting point | 256 to 257 °C (493 to 495 °F; 529 to 530 K) |
| insoluble[1] | |
| Solubility | slightly soluble in ethanol and benzene; soluble in diethyl ether, ethyl acetate and ligroin[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
The compound in the bark gives the tree its white color which appears to protect the tree from mid-winter overheating by the sun. As a result, birches are some of the northernmost occurring deciduous trees.
History
editBetulin was discovered in 1788 by German-Russian chemist Johann Tobias Lowitz.[4][5]
Chemistry
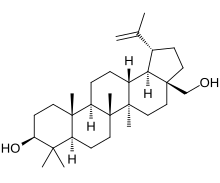
editChemically, betulin is a triterpenoid of lupane structure. It has a pentacyclic ring structure, and hydroxyl groups in positions C3 and C28.
See also
editReferences
edit- ^ a b c Haynes, William M.; Lide, David R.; Bruno, Thomas J. (2014). "3". CRC Handbook of Chemistry and Physics (95th ed.). Boca Raton, Florida: CRC Press. p. 340. ISBN 9781482208689. OCLC 908078665.
- ^ Green, Brian; Bentley, Michael D.; Chung, Bong Y.; Lynch, Nicholas G.; Jensen, Bruce L. (2007-12-01). "Isolation of Betulin and Rearrangement to Allobetulin. A Biomimetic Natural Product Synthesis". Journal of Chemical Education. 84 (12): 1985. Bibcode:2007JChEd..84.1985G. doi:10.1021/ed084p1985.
- ^ Gao, Yuan; Xu, Hongyu; Lu, Zhenming; Xu, Zhenghong (November 2009). "Quantitative determination of steroids in the fruiting bodies and submerged-cultured mycelia of Inonotus obliquus". Se Pu. 27 (6): 745–749. ISSN 1000-8713. PMID 20352924.
- ^ Lowitz, J. T. (1788). "Űber eine neue, fast benzoeartige substanz der briken". Crell's Chem. Ann. 1: 312–317.
- ^ Król, Sylwia Katarzyna; Kiełbus, Michał; Rivero-Müller, Adolfo; Stepulak, Andrzej (2015). "Comprehensive Review on Betulin as a Potent Anticancer Agent". BioMed Research International. 2015: 584189. doi:10.1155/2015/584189. PMC 4383233. PMID 25866796.
🔥 Top keywords: Akademia e Shkencave e RPS te ShqiperiseAlexandria Ocasio-CortezBilderberg GroupCristiano RonaldoDong XiaowanMinecraftOperation GladioPrimal cutRiot FestStrictly Come Dancing (series 7)Main PageSpecial:SearchWikipedia:Featured picturesUEFA Euro 2024Derek JeterJamal MusialaBridgertonInside Out 2UEFA European Championship2024 ICC Men's T20 World CupCleopatraThe Boys (TV series)The Boys season 4Deaths in 2024Pawan KalyanNATO phonetic alphabetUEFA Euro 2020ChatGPTG7Giorgia Meloni.xxxYouTubeNicola CoughlanGermany national football teamBiggest ball of twineOpinion polling for the 2024 United Kingdom general electionJuneteenthJerry WestFlag Day (United States)Project 2025Scotland national football teamJulian NagelsmannDutch rollAndrew McCarthyDonald TrumpThe Acolyte (TV series)ICC Men's T20 World CupFlorian WirtzHit Man (2023 film)